
Very often we are interested in a chemical system under non-standard conditions. Tabulated data for Gibbs free energies are values under standard conditions. Standard absolute entropies of substances are routinely tabulated along with \(ΔH^o_f\) and \(ΔG^o_f\) data.Ĭalculating ΔG Under Non-Standard Conditions Note, that the absolute entropy of an element is not zero, and the absolute entropy of a compound cannot be calculated from the absolute entropies of its elements. Where \(n\) and \(m\) are the stoichiometric coefficients for each product and reactant, respectively. The change in entropy under standard condition for a reaction, \(ΔS^o\), can be calculated from absolute standard entropy data as By the Third Law of Thermodynamics, these values are always positive numbers i.e., \(S_o > 0\). The standard absolute entropy of a substance, \(S_o\), is the entropy of the substance in its standard state at 25 ☌ and 1 atm. However, when using these, it is important to realize that absolute entropies are not changes in entropy, \(ΔS\). 
Values are obtained from the temperature variation of heat capacities. Unlike enthalpy and free energy, absolute entropies, \(S\), can be defined and calculated. 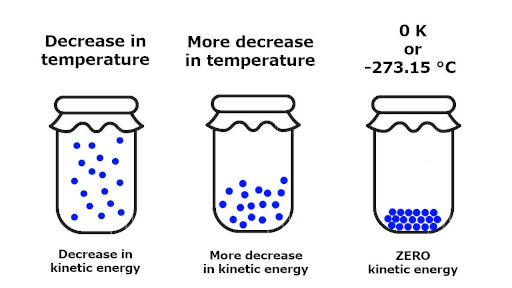 
However, absolute zero is an unattainable temperature and no substance forms a perfect crystal, so all substances have non-zero absolute entropies at all real temperatures. This reasoning lead Walther Nernst in 1906 to formulate what is known as the Third Law of Thermodynamics:Īt the absolute zero of temperature, a perfect crystalline substance would have an absolute entropy of zero. At a temperature of absolute zero we might suppose that a perfect crystal, representing the ultimate order, would have an absolute entropy of zero (\(S = 0\)). If we lower the temperature of a substance, molecular motion will be diminished and greater ordering will occur. With only one possible microstate, the entropy is zero.Depends upon the relative magnitudes of \(ΔH\) and \(TΔS\). Measurements of the heat capacity of a substance and the enthalpies. At temperatures greater than absolute zero, entropy has a positive value, which allows us to measure the absolute entropy of a substance. The third law of thermodynamics establishes the zero for entropy as that of a perfect, pure crystalline solid at 0 K. The third law of thermodynamics states that the entropy of any perfectly ordered, crystalline substance at absolute zero is zero. If \Delta S univ < 0, the process is nonspontaneous, and if \Delta S univ = 0, the system is at equilibrium. The second law of thermodynamics states that a spontaneous process increases the entropy of the universe, S univ > 0. In thermodynamic models, the system and surroundings comprise everything, that is, the universe, and so the following is true: By expanding consideration of entropy changes to include the surroundings, we may reach a significant conclusion regarding the relation between this property and spontaneity. Processes that involve an increase in entropy of the system (\Delta S > 0) are very often spontaneous however, examples to the contrary are plentiful. The third law of thermodynamics has two important consequences: it defines the sign of the entropy of any substance at temperatures above absolute zero as positive, and it provides a fixed reference point that allows us to measure the absolute entropy of any substance at any temperature. In the quest to identify a property that may reliably predict the spontaneity of a process, we have identified a very promising candidate: entropy. Calculate entropy changes for the system, surroundings, and universe for phase transitions and chemical reactions under standard conditions.State and explain the second and third laws of thermodynamics.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
